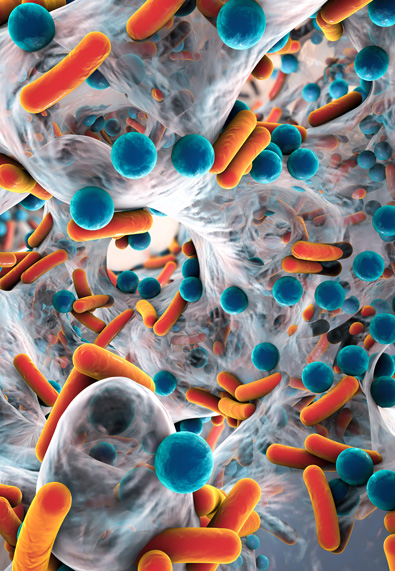
Biofilm Formation and the Increased Risk of a Surgical Infection
Biofilms that remain on or inside of surgical instruments after reprocessing have long been a suspected source of surgical infections. Over the past few years several tragic events have served to prove the link between biofilms on instruments and surgical infections.
One of the more recent, well-publicized events occurred last May at a hospital in Colorado when contaminated surgical instruments posed a problem.1 A month after the initial story made headlines, a report was released that cited biofilms as the probable cause of the contaminated surgical instruments. According to that report, “Biofilm appears to be behind the infection breakout that last month forced the Denver hospital to suspend all surgeries for nearly a week. A trail of evidence points to improper instrument cleaning practices and the hidden menace known as biofilm.”2
The report that came out in June, 2018 pointed out that:
- When surgical site infections are linked back to surgical instruments, biofilm is likely a root cause.
- The infection outbreak spanned nearly 2 years, from July 21, 2016, to April 5, 2018. Whatever was causing the infections were recurrent and undetectable, telltale characteristics of biofilm.
- Colorado Department of Health officials determined that the infection control breach was due to human error that occurred during "a gap" in the manual pre-cleaning phase — before the instruments underwent heat sterilization. Staff wiped down, soaked and scrubbed certain spine and orthopedic instruments, but apparently not well enough. The instruments likely still contained biofilm when they were sent along for automated cleaning and heat sterilization.3
- a) Instruments should be wiped, as needed, with sterile moistened surgical sponges to remove gross soil; and
- b) Cannulated instruments or instruments with lumens should be irrigated with sterile water, as needed, without creating aerosols.5
February 1, 2019
9 view(s) 
